Introduction:
The combination of all-trans-retinoic acid (ATRA) and arsenic trioxide (ATO) has revolutionized the treatment of acute promyelocytic leukemia (APL). However, despite a remission rate exceeding 96% and 5-year disease-free and overall survival rates reaching 96% and 88%, respectively, death during induction in clinical trials occurs in 4% of patients. Differentiation syndrome (DS) is a significant complication during ATRA and/or ATO induction therapy. The signs and symptoms of DS include dyspnea, unexplained fever, weight gain greater than 5 kg, unexplained hypotension, acute renal failure, and the presence of infiltrates and/or pleural/pericardial effusions on chest radiographs. The occurrence of 4 or more of these findings is considered diagnostic. In APL, patients are at risk for fluid overload secondary to multiple blood product transfusions and intravenous hydration. Acute hypoxic respiratory failure (AHRF) and septic shock are common indications for ICU admission in this patient population. At our institution, we monitored patients' daily weights intending to maintain the admission weights with diuresis or intravenous fluids as needed. We assessed the impact of maintaining euvolemia and daily weight monitoring to reduce the incidence of DS and ICU admissions among the APL patients treated at our institution.
Methods:
A retrospective chart review was conducted on 60 patients with APL who received treatment at our institution between 2016 and 2021. A total of 17 patients who were either not induced at our institution or did not have daily weight monitoring were omitted. Patient demographics, median age at diagnosis, hospitalization complications, ICU admission, and DS were analyzed. Patients were grouped based on if their daily weights were maintained at their admission level, plus or minus 3 kilograms. Only standing weights and weights before ICU transfer were considered.
Results:
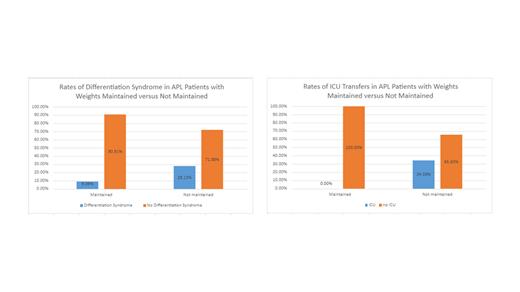
Of the 43 patients who underwent induction therapy at our institution, 25.6% (n=11) had their weight maintained via diuresis and intravenous fluids, and none required ICU admission. Of the remaining 32 patients, 34.4% (n=11) were transferred to the ICU. Of the 11 patients transferred to the ICU, 6 were for AHRF, 2 were for septic shock, 1 was for adrenal insufficiency with vasopressor requirements, 1 was for subdural and epidural hemorrhage, and 1 was for intracranial hemorrhage. Maintaining euvolemia and weight within 3 kilograms of admission level correlated with fewer ICU admissions (OR = 0.085, 95% CI = 0.0046-1.5761) (p=0.0980). Of the 11 patients whose weights were followed and maintained, 9% (n=1) developed DS. Of the 32 patients who did not have their weights followed and maintained, 28% (n=9) met the criteria for DS (OR = 0.256, 95% CI = 0.028-2.296) (p=0.223).
Conclusion:
Volume overload is a significant concern in APL and its treatment. Our findings suggest that maintaining euvolemia and daily weight monitoring can lead to effective management of patients on the floor and may prevent unnecessary ICU transfers. Although there are limitations to the study due to its retrospective nature and low sample size, the cost-effectiveness and noninvasive methods of maintaining euvolemia support its potential addition to the standard treatment protocols. While maintaining euvolemia and monitoring daily weights is a simple intervention, it represents a promising strategy to improve the management of APL patients during induction therapy by decreasing the incidence of DS and ICU admissions. Further prospective studies are warranted to validate and expand upon these findings.
Disclosures
Keruakous:BMS: Membership on an entity's Board of Directors or advisory committees; Janssen: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding. Cortes:Pfizer: Consultancy, Research Funding; Abbvie: Consultancy, Research Funding; Forma Therapuetic: Consultancy; Gilead: Consultancy; Takeda: Consultancy, Honoraria; Biopath Holdings: Consultancy, Current holder of stock options in a privately-held company, Membership on an entity's Board of Directors or advisory committees, Research Funding; Novartis: Consultancy, Research Funding. Kota:Pfizer: Honoraria; Kite: Honoraria; Incyte: Research Funding; Novartis: Honoraria.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal